|
3/24/2024 0 Comments Bohr model of the atom definition The quantum mechanical model specifies the probability of finding an electron in the three-dimensional space around the nucleus and is based on solutions of the Schrödinger equation. The Quantum Mechanical Model: Definition & Overview. 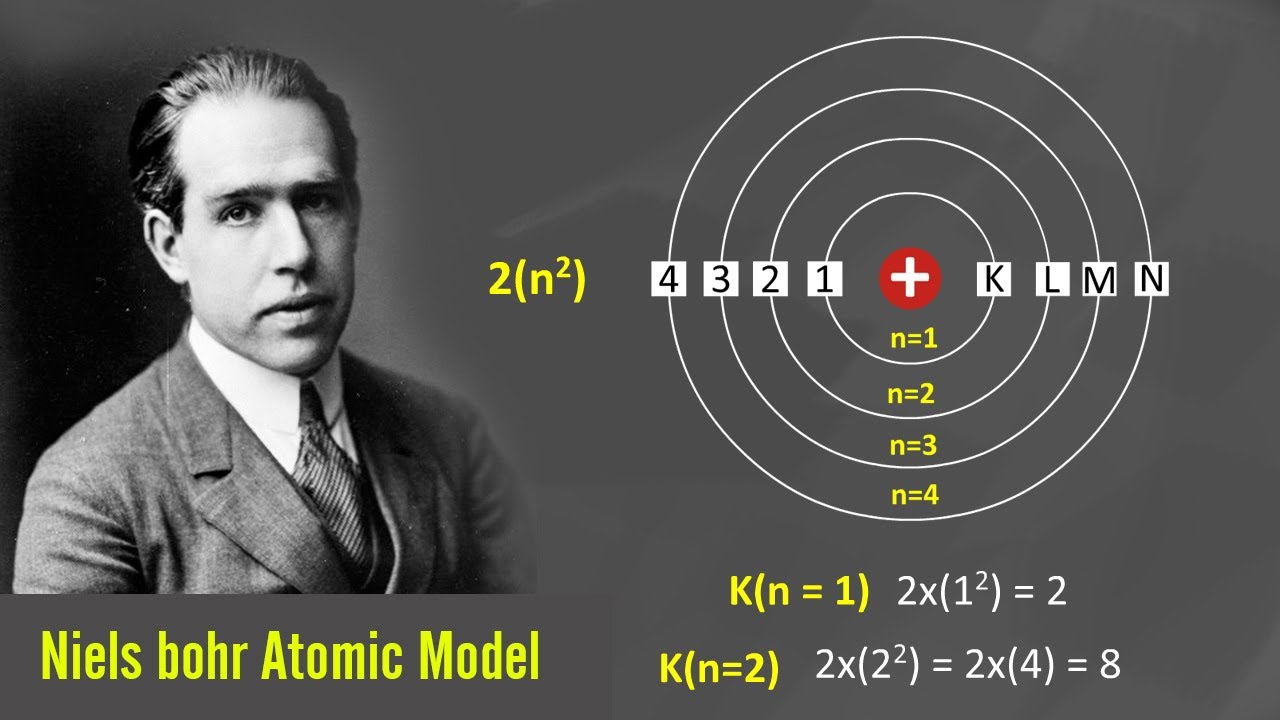
Encyclopædia Britannica, inc., 5 June 2014, Available here. As a theory, it can be derived as a first-order approximation of the hydrogen atom using the broader and much more accurate quantum mechanics and thus may be considered to be an obsolete scientific theory. An atomic orbital, which is distinct from an orbit, is a general region in an atom within which an electron is most probable to reside. Bohr Model: Bohr model is an atomic model that was proposed by Niels Bohr (in 1915) to explain the structure of an atom. Instead of allowing for continuous values for the angular momentum, energy, and orbit radius, Bohr assumed that only discrete values for these could occur (actually, quantizing any one of these would imply that the other two are also quantized). The Bohr atomic model, proposed by Niels Bohr in 1913, was a theory that revolutionized our understanding of atomic structure. The Bohr model is a relatively primitive model of the hydrogen atom, compared to the valence shell model. The absolute value of the energy difference is used, since frequencies and wavelengths are always positive. The electron’s speed is largest in the first Bohr orbit, for n 1, which is the orbit closest to the nucleus. 
In this equation, h is Planck’s constant and E i and E f are the initial and final orbital energies, respectively. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
